Infection Sleuths: Deciphering the links of subversion
How scientists can leverage scRNA-seq to understand the "tricks up the sleeve" of infective microbes.
Dear Community,
I was pondering on what to write for the lovely folks of this community. This week marked international women’s day and I wanted to write something that touched on an aspect of womanhood. It was slightly hard to find a journal article that had an intersection of applied bioinformatics and understanding a physiological process. Luckily, There was a beautiful work recently published in EMBO MSB by Itai Yanai’s team. This was also accompanied by an awesome cover image by the artist Emma Vidal’s interpretation of Hope II by Klimt. Klimt along with Vermeer and Caravaggio are a few of my favourite artists, So I had to dig through the paper.
This story (article) is quite a ride, It demonstrates how we could come up with a hypothesis/observation and test using an animal model and further using scRNA-seq to untangle the mystery.
Before we get into the details;
Please leave me comments and critiques; that’s the only way I can improve and deliver value to you.
It is important for me that you are enjoying the read, but also gaining insights and growing your knowledge.
If you can tweet about the newsletter, It would be awesome. This is a community and we are here to support each other, spreading the word will attract valuable comments and insights.
Origins
The origin of mammalian life and its incubation period often occurs in an extra-embryonic organ only present during pregnancy and the anatomical boundary between the mother and the fetus; The placenta is an organ of wide-ranging functions. As a physical barrier between maternal and fetal circulation; It limits the transfer of pathogens. It also provides a space for nutrient transfer, gas exchange and hormonal production. It is also a centre for defence against pathogens that have wandered into the sanctuary with “malicious intents”. Alternative routes such as the intervillous space, the implantation site and the fetal membranes are the frequent sites of vertical transmission, these sites are usually in direct contact with the maternal biological systems. Pathogens tend to manoeuvre through these sites and cause infections in the fetus which in turn can be deadly.
The defence centre employs a variety of small molecules and cells to perform the function of keeping pathogens at bat while the fetus is gently rocking in the placenta growing and its cells and organs being nurtured by the mother. To limit the ascending infection from pathogens and pathobionts present in the lower genital tract, mechanisms like the production of mucus (limits mobility and traps the pathogen) and antimicrobial peptides are produced in the cervix. This is one of the mechanisms of a plethora that the defence centre employs. The other more competent warriors are the components of the innate immune system.
Cellular life in the placenta
The types of cells as you journey through the placenta are vast, and the placenta is the first and largest organ that the fetus makes. The placental villi also called chorionic villi sprout from the chorion to provide a contact area with the maternal blood. These are essential elements in pregnancy and are the product of conception. Umbilical arteries carry embryonic blood to the villi and post circulation the blood returns to the embryo through the umbilical vein.
The placental villi are composed of three layers of components with different cell types in each.
Syncytiotrophoblasts cover the entire surface of the villous tree and bathe in the maternal blood within the intervillous space.
Mesenchymal cells, Mesenchymal derived macrophages (Hofbauer cells) and fibroblasts. They are located within villous core stroma between trophoblasts and fetal vessels
Hofbauer cells synthesize VEGF and other proangiogenic factors that initiate vasculogenesis (the process of forming new blood vessels).
HB have a variety of functions including control of villous remodelling and differentiation, and hormonal secretion and may have a role in infection during pregnancy.
Fetal vascular cells include vascular smooth muscle cells, perivascular cells and endothelial cells.
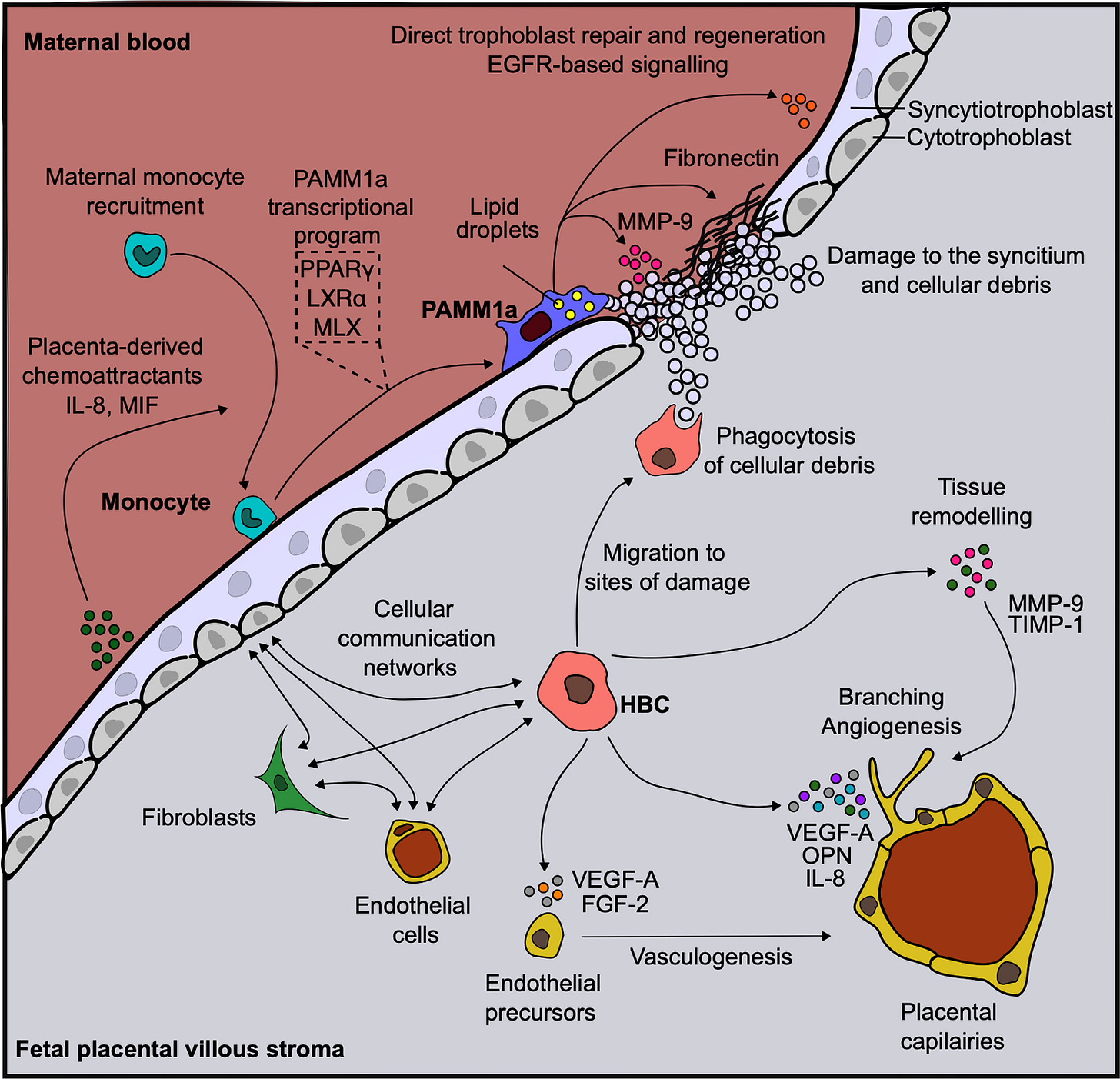
Host inflammatory dynamics reveal placental immune modulation by Group B streptococcus during pregnancy.
Group B Streptococcus(GBS; a group of common bacteria) is one of the main causes of neonatal septicemia (blood infections) and meningitis. GBS behaves as a commensal organism that colonizes the lower GI tract and can traverse defined anatomic barriers like the cervical and vaginal epithelium. GBS strains are encapsulated with specific capsular polysaccharides which are then the virulence factors and they stimulate antibody-associated immunity. The invasion of GBS involves the attachment of the streptococcal chains to the plasma membrane of the cells (such as Amniotic epithelium) and engulfment of the adherent bacteria by cellular protrusions and formation of membrane-bound vacuoles; Essentially the bacteria get attached to the cell and absorbed in a balloon like structure.
Macrophages are one of the immune cells within the placenta; Their origins can be either maternal or fetal and as mentioned above play a diverse set of roles and harmonious synchronization of these functions are important for a successful pregnancy. These macrophages also balance the pro- and anti-inflammatory nature and the balance has been associated with the outcomes of pregnancy. In a GBS infection, the role of these macrophages and how they contribute towards the pathological outcome is not clearly understood. Amongst the toxins produced by GBS, ß-hemolysin/cytolysin are pore-forming toxins that lyse erythrocytes (red blood cells) that work by attacking the bilayer membranes and destroying the phospholipids present there.
Hemolysins are secreted by the bacteria and diffuse through the water present in their surroundings before they are picked up by specific receptors where they organize themselves into a ring-shaped complex; It basically creates a hole so the contents of the cell are disrupted by the release of organic phosphorus from sphingomyelin and this causes cell lysis.
In the current paper:
They defined placental response to GBS infection by measuring phenotypic changes in the tissue and changes in RNA expression(transcriptomic) in the cells.
The experiment was done in a time-course manner to monitor the progressive changes.
By focussing on the role of ß-h/c in the modulation of innate immune response they try to address how important this interaction is to different outcomes.
They propose that the findings in the article reveal GBS immune evasion strategy and cause infection by controlling the placental innate immune response.
If you made it untill here, Yes there is a bio-informatics component to this paper and we will go through it and see how the authors made informed decisions based on the scSEQ data.
Climbing through: the capture of differences in response over time.
Modelling infections are often the common approach as you cant deliberately infect a human being; scientists use a variety of models ranging from in-vitro cell culture methods to animal models that could potentially give us insights into what is happening in humans. The authors of the study used mice and administered wild type or KO GBS(lacking ß-h/c) or sham (nothing but the carriers of the infection agent, in this case, 1:1 PBS: Gelatin(10%)) post-day 13 of pregnancy. Following these administrations, they conducted a time series experiment where the mice were euthanized at 12h, 24h, 48h, and 72h corresponding to E13.5 till E16 of the embryonic life cycle.

Key observations
By measuring the bacterial burden(amount of bacteria per gram of placenta) it was observed that bacteria expressing the toxins had a higher burden vs the ones not expressing toxins.
Confirmed it with immunofluorescence and histological staining.
The bacterial load increased depending on the time point; ie there are more CFU (colony-forming units) as the days of infection progressed.
They observed an increase in macrophage staining in the heavily colonized tissues, so there were some interactions between the macrophages and the toxins going on.
So if you were given this challenge to understand what mechanisms or even cells respond to an infection at the tissue level what likely experiments would you come up with (leave a comment below). On my part, If I am restricted from reaching transcriptomics, I would likely use staining for different cell types and flow cytometry to address such challenges. But with access to scSEQ, these slightly tedious procedures can be skipped. Which is what the authors did.
Leveraging the granularity that can be obtained with scSEQ experiments, the authors performed single-cell sequencing of samples post 48h infection. This was due to the severe phenotype after 48h of infection.
They found that cell types contributed to the transcriptional variance among the samples.
They identified trophoblast, immune and erythrocyte cell lineages based on markers distinct for them.
zooming in trophoblast and immune lineages (aka sub clustering) revealed the presence of various cell types.
By comparing cells within each lineage from sham and GBS-infected mice they calculated Pearson’s correlation which showed similar distributions among erythrocytes and trophoblasts but the immune lineage was less correlated. This gives the idea that a large influx of immune cells might be the immune cells that are present in a steady state in the placenta.
They also checked if subclusters underwent changes in transcription ie., do they participate in the process of infection response? By using Differential gene expression they show that there is an enrichment in inflammatory genes in GBS-infected erythrocyte lineage cells
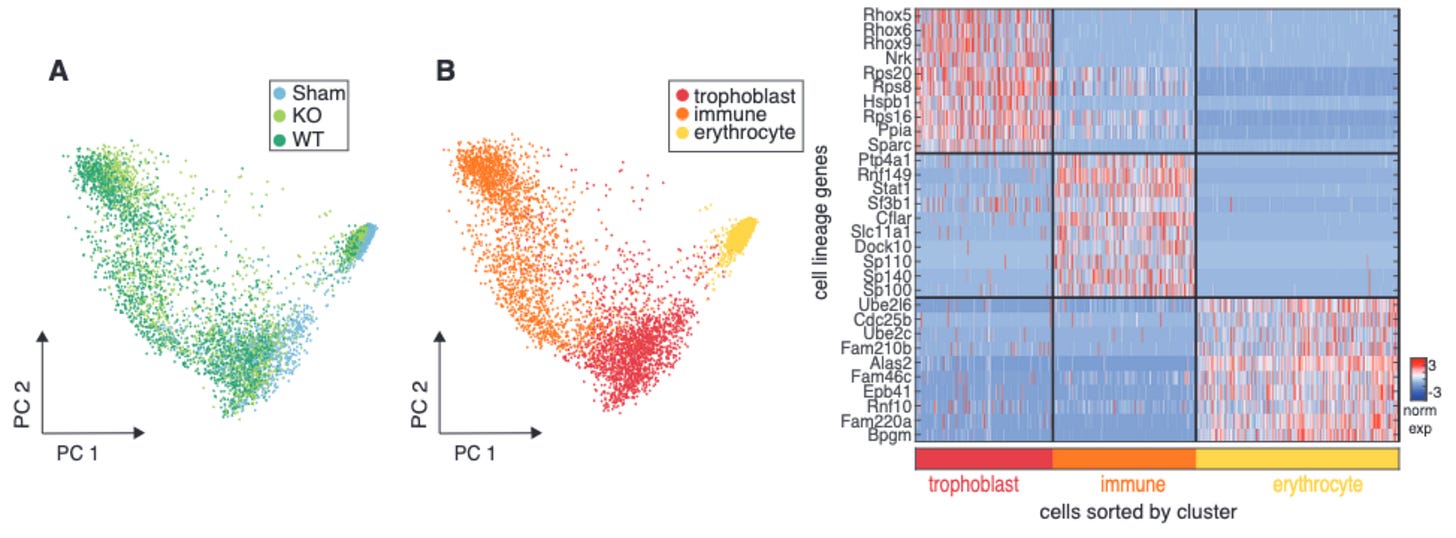
Okay, but erythrocytes arent “immune” cells right, yes they are not but they deep dive into the immune cell profiles and here is the breakdown.
Based on the principle components of the neutrophil profile they found that in the early phase post infection neutrophils from WT-infected mice have higher inflammatory expression than the matched KO-infected mice
In severe phenotype, there is a shift in this inflammatory expression. The KO-infected mice actually have higher inflammatory gene expression than those of WT even if this is not clearly apparent at the tissue level phenotype.
Interleukin-1 family of inflammatory response in neutrophils are induced by both the trains but the response is lower in WT.
This kind of hints that WT has some advantageous mechanisms and by subverting inflammation response by neutrophils they are able to “incubate” longer.
But I did mention that macrophages are present around, what about them you should ask:
like neutrophils, the peak macrophage response was observed at 72h post-infection.
PCA was used to distinguish cells of maternal and fetal origin. They confirmed the origins using cells expressing Y-chromosome genes would not be from the mothers, especially in cells from male fetuses.
They further wanted to check whether the two different origin cells were affected by GBS infection since they are important in mediating placental functions.
They found the fetal macrophages from WT-infected placentas at 73h are the only cells that exhibit inflammatory genes. This was based on differential expression.
They checked the response of maternal macrophages between conditions after 72 hours of infection. Macrophages from ß-h/c-KO infected mice had higher PC-1 scores and showed the expression of inflammatory response genes.
So, the cells from WT-infected placentas show greater inflammatory expression relative to KO-infected placentas at the first stage (up to 48h), but this switches around after 48h of infection for the maternal macrophages. The fetal macrophages do not show an inflammatory response until the point of severe phenotype, and only WT-infected cells express inflammatory genes in the fetal macrophages.

The trend here seems that the toxins are playing a role in modulating the dynamics of inflammatory gene responses in both the neutrophils and macrophages. To test this in the human placental macrophage population; They obtained cells from three full-term human placentas and cultured the macrophages from them. These cultured macrophages were stimulated with KO or WT GBS.
Using tSNE they were able to see two clusters corresponding to placental macrophages and fetal macrophages(HB cells introduced above).
One of the replicates was of male origin, they used the Y chromosome gene expression to validate the fetal origin of the macrophages.
Since the most number of cells that were responding to the stimulation was from the maternal origin, they focussed on the maternal macrophage subset.
By comparing the inflammatory programs they find that inflammatory gene expression from KO-infected macrophages was significantly higher versus WT.
This led to their hypothesis that the presence of ß-h/c suppresses inflammatory response in the maternal intrauterine compartment.
By using PC scores analysis, they find that KO-infected macrophages were significantly enriched in IL-1-inflammatory response genes. Similarly, at a protein level IL6, CCL3, TNFα, CCL4, CXCL1 etc were significantly higher in KO than in WT.
Note: These are the genes that play a significant role in infection response and clearance.
Whereas in the WT GBS infection, they see enrichment of genes involving regulation of inflammation in other physiological contexts.
This suggests that after the initial inflammation response to GBS, the toxins could actively suppress the inflammation through the induction of suppressive and regulatory genes.

Summary:
GBS infections are still prevalent and contribute to pre-term fatalities, till now we did not have a clear picture of how these bacteria modulate immune responses.
With increasing antibiotic resistance, We might face worse situations towards clearing the infections. So coming up with an alternative targetted approach is crucial.
In this case, it seems that the toxins subvert the inflammatory response during late infection.
This leads to the destruction of barriers and inflammation of membranes.
This is a beautiful story, I can’t stress the elegance of the experiments. As I mentioned in a previous post, They did an excellent job as sleuths gathering evidence and confirming their hypothesis. I also love the simplicity of the model which has provided insights into the infection process. Being trained in Infectious diseases before switching to bioinformatics, these sorts of papers get me excited and make me revisit older experiments and studies with a new lens.
References:
Wang Y, Zhao S. Vascular Biology of the Placenta. San Rafael (CA): Morgan & Claypool Life Sciences; 2010. Chapter 4, Cell Types of the Placenta. Available from: https://www.ncbi.nlm.nih.gov/books/NBK53245/
Kuperwaser F, Avital G, Vaz MJ, Noble KN, Dammann AN, Randis TM, Aronoff DM, Ratner AJ, Yanai I. Host inflammatory dynamics reveal placental immune modulation by Group B Streptococcus during pregnancy. Mol Syst Biol. 2023 Mar 9;19(3):e11021. doi: 10.15252/msb.202211021. Epub 2023 Feb 6. PMID: 36744393; PMCID: PMC9996236.
Thomas JR, Naidu P, Appios A, McGovern N. The Ontogeny and Function of Placental Macrophages. Front Immunol. 2021 Oct 21;12:771054. doi: 10.3389/fimmu.2021.771054. PMID: 34745147; PMCID: PMC8566952.
Socials:
Please follow or check the labs linked here, supporting them is supporting scientific community:
Felicia Kuperwaser: Twitter
Cover image: Emma Vidal
Cover Image
If I ever make it to a cover image I am sure Emma will be the person who I will contact as the artist.
Supporter gratitude:
Thank you Gendy, for supporting this article with a cup of coffee. I highly appreciate your support and contribution.
Disclaimer:
I am not an expert in this field so if there are errors please contact me so I can correct them ASAP.
If you enjoyed the article, Please leave a like and comment below. And if you appreciate the work, maybe scan the QR code and buy me a coffee.
See you next week with a cool new paper on a scSEQ technology that is going to change the field.







